Endress+Hauser highlights growing trends within the process automation industry
Discover the top 10 most viewed blogs of 2022 covering challenges and solutions across all industries May 9, 2023 – […]
The ever-looming hydrogen economy has found renewed life in the energy transition as industry leaders in public and private sectors find a sense of urgency in decarbonizing our industrial economy. Hydrogen is a clean burning molecule and can be produced without emissions. This is done either through traditional Steam Methane Reformation (SMR), gasification processes with carbon capture, or by electrolysis which takes advantage of low cost or excess renewable energy in our electric grid.
For hard to electrify thermal loads, “green-ing” refineries and chemical processes, hydrogen emerges as the best candidate for a net zero economy.
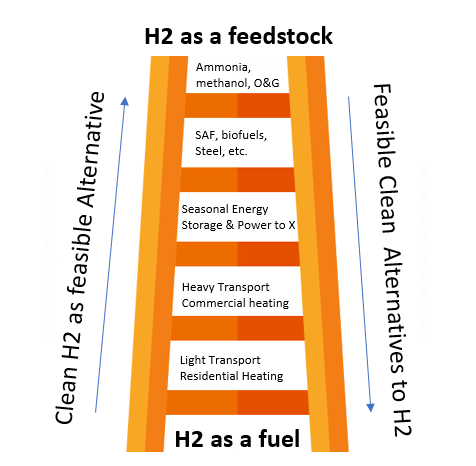
There is an active, rigorous debate on how to prioritize the use of this molecule as it is still expensive to produce relative to traditional or alternative fuels and energy carriers, but it is unavoidable in the context of a carbon neutral society.
By 2050, hydrogen production is expected to make up 12% of the world’s energy supply with a wide range of novel applications like blending hydrogen in Natural Gas or Utility Scale Electrolyzers (IRENA).
Endress+Hauser manufactures process instrumentation that is used across the full value chain of hydrogen from production to end use.
With a long history of counsel and participation in hydrogen processes, Endress+Hauser can offer specific guidance on the evolving challenges presented to instrumentation in this re-emerging market. One critical measurement, pressure, faces a unique risk: Hydrogen permeation of the process membrane.
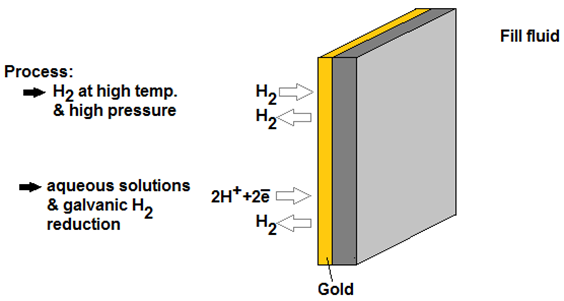
In direct hydrogen gas service, hydrogen molecules dissociate on the diaphragm surface of the pressure transmitter. Next, hydrogen atoms lose electrons and the subsequent H+ ions diffuse through standard 316L or Alloy C diaphragms. Once on the other side of the metal diaphragm, the H+ ions capture electrons and recombine into H2 molecules. These new H2 molecules then go into solution in the pressure cells fill fluid.
The speed and degree that permeation occurs will be impacted by the process pressure and temperature.
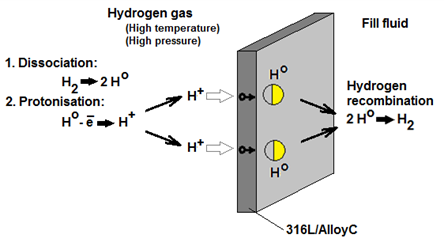
The danger from hydrogen permeation is not immediate. So long as sufficient process pressure is maintained on the diaphragm, the H2 molecules will remain in solution. It is only when the process pressure drops to near or below atmospheric pressure that damage occurs. With reduced process pressure, H2 molecules in the fill fluid come out of the solution and form gas bubbles. This happens very quickly, increasing the fluid volume and internal pressure. Since the pressure cell or diaphragm seal is a closed environment, this additional volume has no means of escape. Consequently, the thin 316L / Alloy C membrane becomes distended, resulting in catastrophic damage.


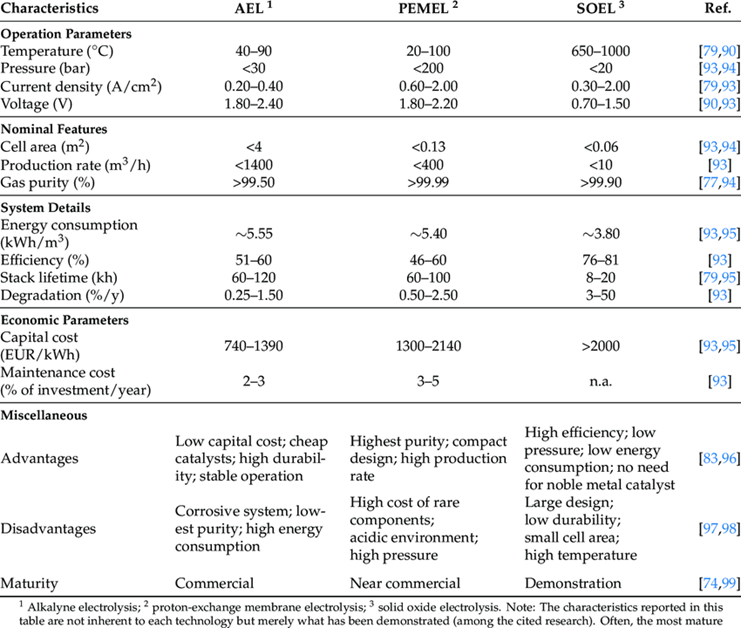
There are three emerging technologies for large scale electrolysis, which is the process of splitting H2O with high DC currents into its constituents H2 and O2.
Pressure is an important parameter as many of these technologies operate based on differential pressures across the membrane. After the H2O is converted, the hydrogen gas is treated/dried for the eventual end use. Outlet pressure or differential pressures across critical components are important indicators of a healthy process and in some cases used for Safety Instrumented Systems (SIS) or functional safety requirements.
As such, the risk of hydrogen permeation in pressure transmitters is ever present in these new production techniques with various process conditions which directly impact the risk of hydrogen permeation through pressure transmitter diaphragms.
There are more complicated matters at hand, deviating from the typical hydrogen applications of the past. As seen in recent power plant demonstrations, such as Long Ridge Energy’s Hydrogen Ready Power Plant in Hannibal, Ohio, there is a growing interest in blending hydrogen into natural gas to reduce the carbon content in your gas pipelines or fuel systems.
The image below represents the pipeline application. Compressors are used to inject blended gas back into the pipeline. It is critical to keep the injection pressure higher than the pipeline pressure to ensure the blended gas will be pushed into the pipeline. Any pressure transmitters on the blended gas line and downstream of this injection site would be subject to some increased percentage of hydrogen by volume. Failures in pressure measurements could cause a potential trip in the control loop if the supply pressure faults.
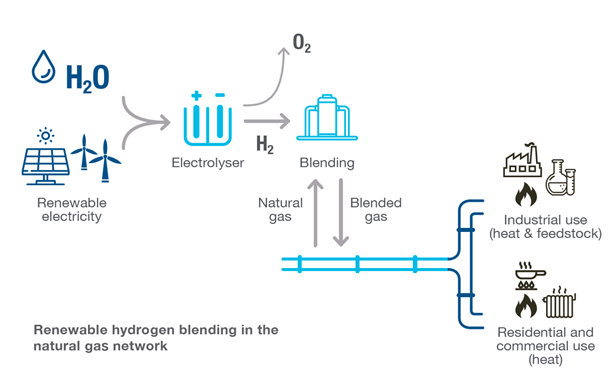
The risks of permeation are different at various blends of hydrogen, offering further complexity to end users or engineering firms designing new systems or validating the existing infrastructure’s capabilities relative to the potential percentage of hydrogen by volume.
The most common approach is to employ a gold [Au] coating on the external face of the diaphragm. This gold layer increases the density of the diaphragm thus creating a diffusion barrier to impede hydrogen permeation. Properly utilized, gold coating will decrease hydrogen permeation up to 106 times due to a very low diffusion coefficient.
There is a wide variety available but not all are suitable for hydrogen permeation prevention. A nominal coating thickness of 25 µm strikes a good balance between protection and cost. A thinner coating is more difficult to apply evenly and risks the development of pinholes and reduced protection. Gold coatings >25µm in thickness are also available with associated increase in cost and lead time.
Hydrogen permeation in conjunction with metal diaphragms is relentless. Consequently, no coating will prevent it from occurring. However, properly employed, gold coating is a cost-effective method to significantly reduce hydrogen permeation and extend the life of your pressure transmitter.
Reduction in the volume of hydrogen present also reduces but doesn’t eliminate the risk of hydrogen permeation. The most conservative design approach, even with blended applications, is to utilize gold coated membranes.
If hydrogen blending will be temporary or < 10% by volume, you may consider employing traditional 316L stainless steel membranes.
Citations:
Discover the top 10 most viewed blogs of 2022 covering challenges and solutions across all industries May 9, 2023 – […]
Company continues commitment to hydrogen safety, with a focus on green hydrogen April 18, 2023 – Given measurement’s critical role […]
With the help of process measurement solutions from Endress+Hauser, a new Long Ridge Energy (LRE) power plant is successfully showcasing […]