Instruments are vital in process environments because their measurements directly impact product quality and operational safety. However, even the most accurate instruments exhibit a certain degree of measurement error, which is the difference between the measured and a reference value.
The magnitude of error differs depending on the type, function and conditions under which an instrument is placed. Consequently, a general requirement exists to regularly quantify these errors and to determine whether measurements are reliable enough to serve their intended purpose.
Calibration and verification defined
Calibration is a procedure for establishing a relationship between a quantitative measurement and a known reference. Verification is typically a qualitative provision of objective evidence that a given measurement fulfills specified requirements.
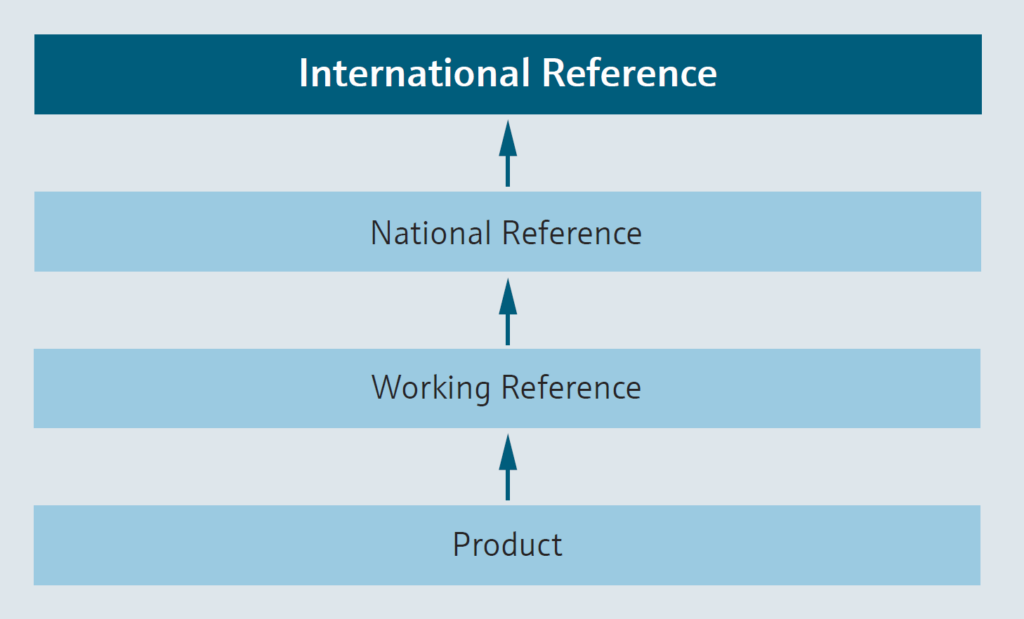
The goal of calibration is to determine the measurement errors of a given device, referred to as the unit under test (UUT). Once the measurement errors become known, a UUT can be used as a reference to calibrate another instrument. A valid chain of calibrations must always lead back to a national or international primary reference standard to create a guarantee of metrological traceability.
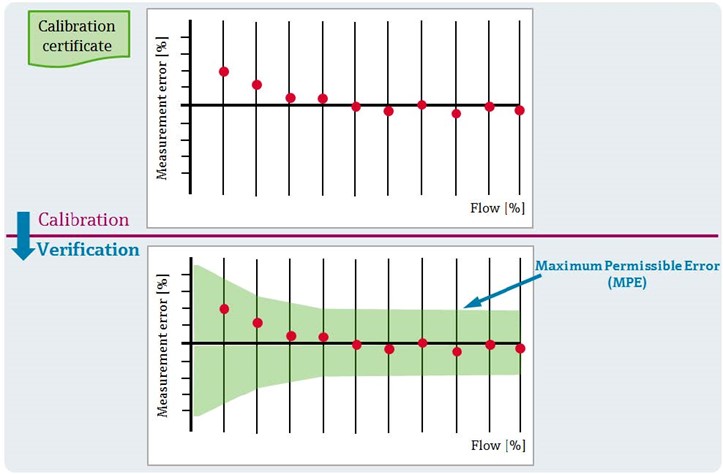
The most common requirement used for calibration is maximum permissible error (MPE). This is defined by a manufacturer, a metrology organization, an end user, or an industry regulatory authority. If a device’s measurement errors are less than or equal to the MPE, then the instrument fulfills the specified requirement and passes its calibration test.
The MPE, combined with regulatory requirements, is often used to determine calibration frequency and procedures.
Instruments with a strict MPE must often be lab-tested under stringent conditions. However, evaluators can frequently make the case for less disruptive calibration methods—or even inline verification—for instrumentation with less strict MPEs.
The concept of verification is more comprehensive . It can be accomplished in a similar manner as calibration but would typically be accomplished with a reference which does not meet the metrological traceability requirements of a calibration. For instance, this can occur in using a clamp-on ultrasonic flowmeter to verify an inline flowmeter. Another common way is to test the various components, which are part of the measurement signal, to ensure that they are of good working quality. As an example, this could be accomplished in an electromagnetic flowmeter by testing the electrode voltages either with an internal or external reference.
For instruments that support it, such as Endress+Hauser instrumentation equipped with Heartbeat Technology, inline verification improves plant availability and cuts costs. This is accomplished by reducing calibration frequency, risk of damage during transit and decrease in mistakes during instrument reinstallation.
Additionally, this more frequent and easily accessible method for verification empowers operators to track an instrument’s performance over time, providing early notice of measurement drift. This information can be used to reduce unscheduled downtime.
Extend calibration intervals using verification
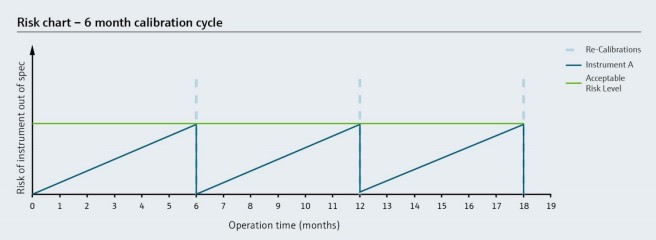
A calibration interval, or the time between calibrations, should be determined in alignment with instrument criticality and the risk of unacceptable measurement error. This risk of error increases over time, but regular calibration helps keep it at an acceptable level. However, full-scale calibration is often time-consuming and costly.
Calibration alone provides a direct measurement of instrument error, but verification after calibration puts the result in regulatory context, helping plant personnel ensure instrument performance remains in compliance.
Verification can also be performed between calibrations, which can extend the calibration interval in many cases. This is especially convenient for instruments that support inline verification, which is achieved by leveraging onboard diagnostic data to perform regular health checks.
Many of today’s instruments possess these built-in capabilities to diagnose problems, perform verification and generate auditable reports. These capabilities ease both calibration and verification-related tasks which reduce intervention and plant downtime. This approach also ensures traceability, which is required in all certified calibration and verification procedures.
Drive process improvement
A process manufacturers’ competitive edge depends on accurate measurement and efficient production. To ensure this is possible, it is critical to understand the relationship between calibration and verification.
The approach outlined in this blog post empowers plant personnel to increase instrument efficiency, achieve maximum uptime, maintain regulatory compliance and uphold the highest degree of safety.
For further questions regarding topics discussed in this blog, please fill the form out below to speak with one of our experts: